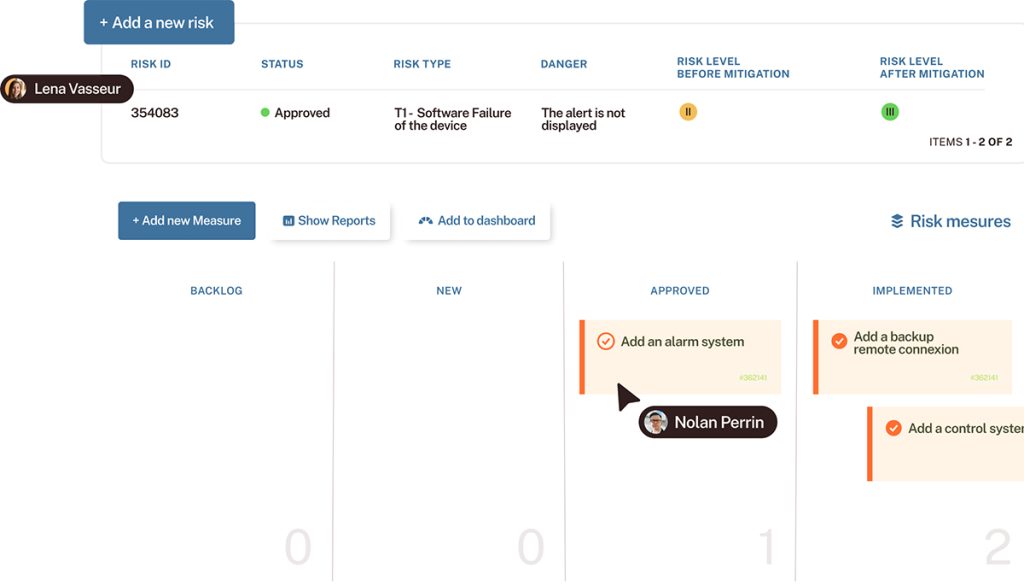
In the MedTech industry, to market your devices or software applications, you must prove that your development and quality management processes are properly managed. Are you ready?
Can you demonstrate that your processes are under control, with a traceability from A to Z to eliminate errors? Is it easy for your teams?
In this webinar, we will answer a question that could be asked during a compliance audit. We will show how our clients prove to auditors that their processes are handled: from requirement management, to tests, to code versioning to patches in case of issue.